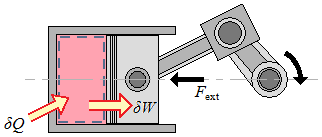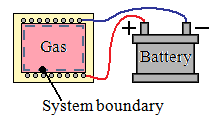
Only the gas is considered as the system. The energy crosses the boundary of the system because the temperature of the walls is higher than the temperature of the gas. Therefore, heat crosses the boundary of the system.
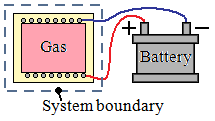
Here, the system includes the vessel and the resistance heater. Electricity crosses the boundary of the system, this is work.